SSV Farmatsevtika mahsulotlari xavfsizligi markazida Farmakologiya qo‘mitasi raisi o'rinbosari Maxfuza Amirova boshchiligida qo‘mita Ilmiy va ekspert kengashining 2-sonli majlisi bo‘lib o‘tdi.
More

SSV Farmatsevtika mahsulotlari xavfsizligi markazida Farmakologiya qo‘mitasi raisi o'rinbosari Maxfuza Amirova boshchiligida qo‘mita Ilmiy va ekspert kengashining 2-sonli majlisi bo‘lib o‘tdi.
More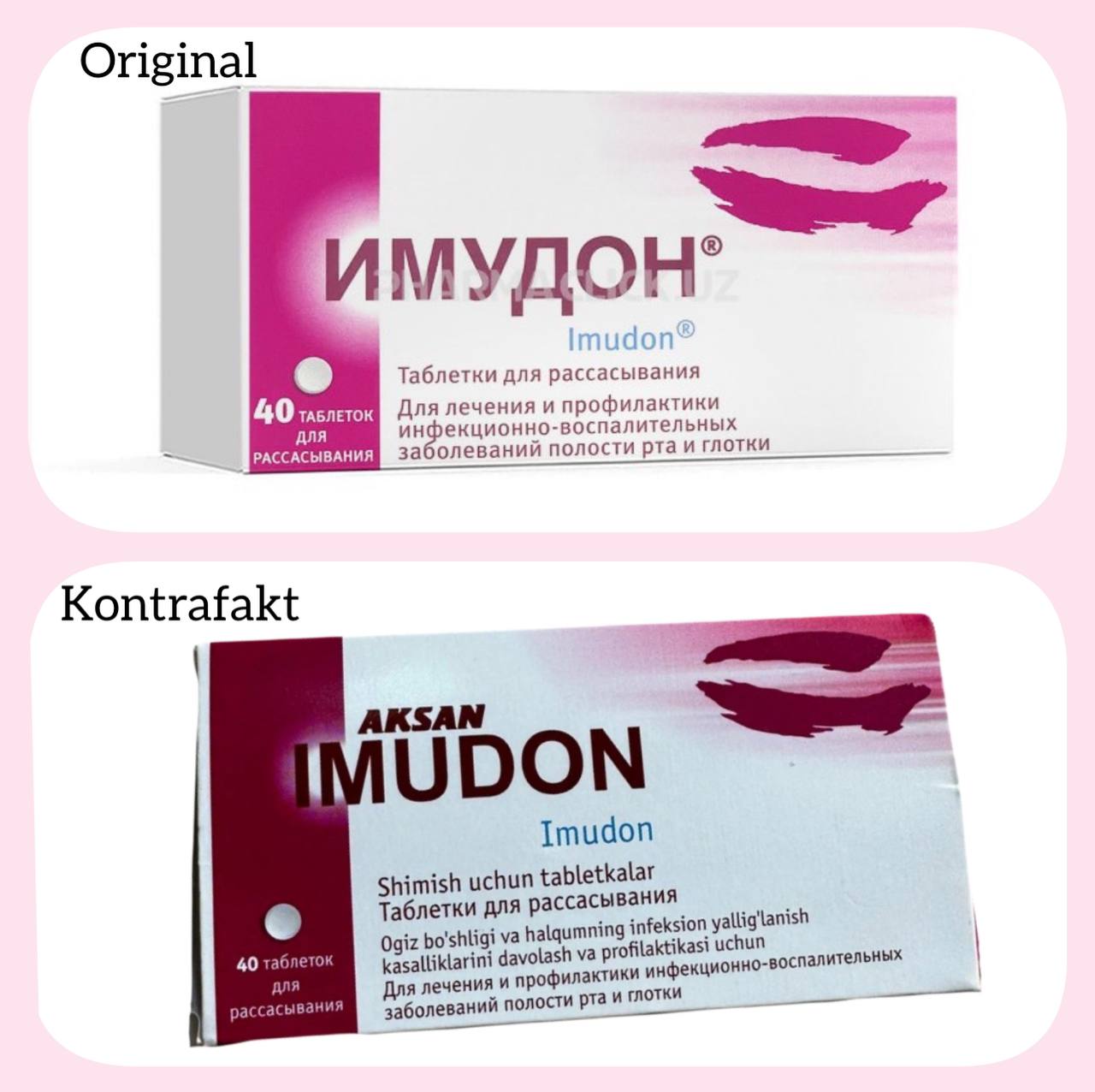
O‘zbekiston Respublikasi Adliya vazirligi xabariga ko'ra, “Abbot Products Operations AG” (Shvetsariya) kompaniyasining vazrilikka yo'llagan murojaatida, unga tegishli bo‘lgan “IMUDON” tovar belgisidan noqonuniy foydalanish holati yuzasidan tegishli choralar ko‘rish so'ralgan.
MoreIjtimoiy tarmoqda keng muhokamaga sabab bo‘lgan, "Faringo forte" spreyining ishlab chiqarish jarayoni aks etgan video bo‘yicha Sanepidqo‘mita mutaxassislari ishlab chiqaruvchi tadbirkor bilan bog‘lanib, mavjud hujjatlarini o‘rganib chiqdi. (https://telegra.ph/file/048351724767ac455db66.png)
MoreDori vositasi ekanligi aytilayotgan “Farengoforte” savdo nomidagi mahsulot aslida dori vositasi sifatida davlat ro’yxatidan o'tkazilmagan va tibbiyot amaliyotida qo’llash uchun ruxsat berilmagan.
More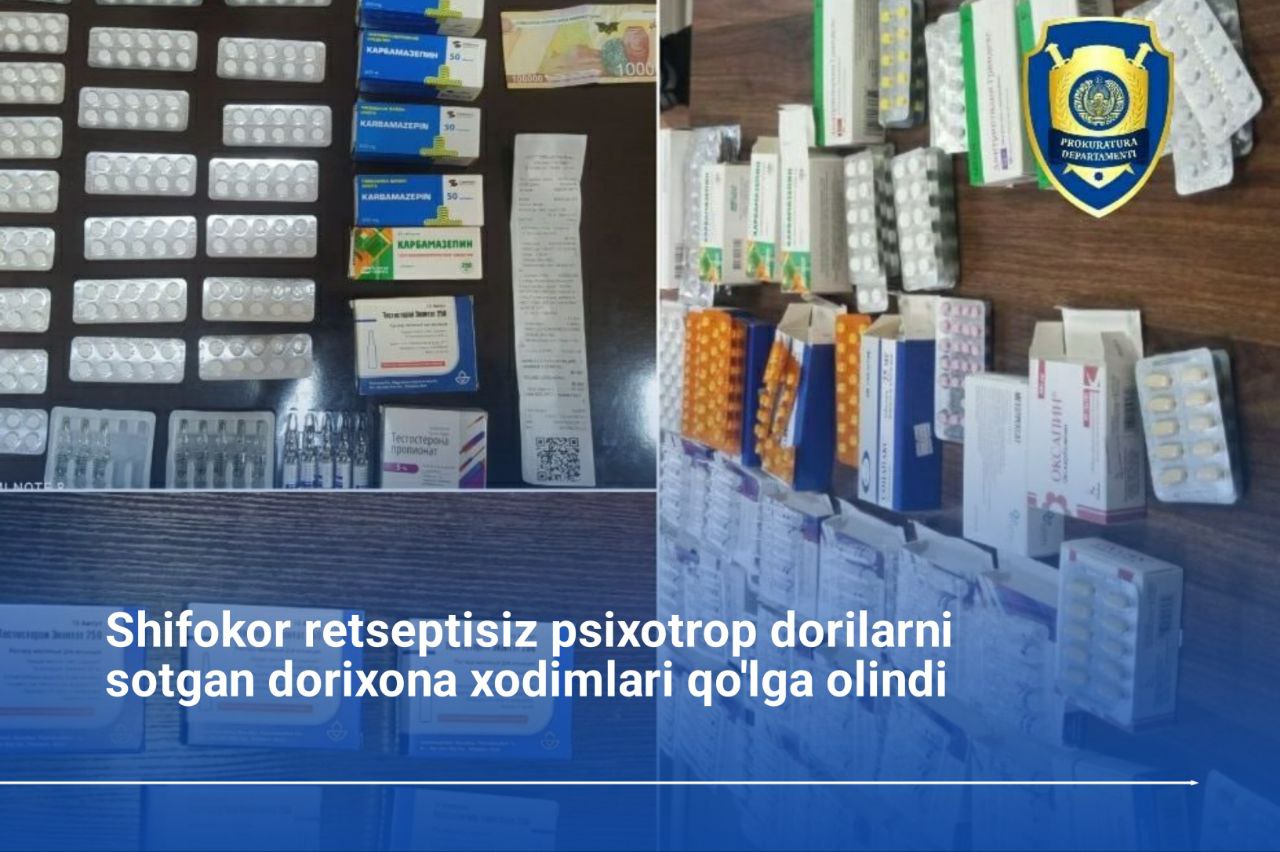
Bosh prokuratura huzuridagi Departamentning Denov tuman bo‘limi tomonidan o‘tkazilgan tezkor tadbirda, fuqaro S.F. jami 40 dona “Regapen” nomli kuchli taʼsir qiluvchi dori vositasini fuqaro A.A.ga 3,8 mln so‘mga noqonuniy sotgan vaqtida ashyoviy dalillar bilan ushlandi.
More
Bundan kelib chiqib, demak, har doim preparatlarni kerak vaqtida dorixonalardan sotib olish kerak ekanda, degan fikr uyg‘onmasligi lozim. Sababi har bir uyda dori qutisi bo‘lishi tavsiya etiladi.
MoreKelgusida etilenglikol va/yoki dietilenglikol bilan kontaminatsiyalangan dori vositalarining qabul qilinishi natijasida kuzatiladigan noxush holatlarni oldini olish maqsadida, Sog'liqni saqlash vazirligi huzuridagi "Farmatsevtika mahsulotlari xavfsizligi markazi" davlat muassasasi direktorining tegishli buyrug’i qabul qilingan.
More
Bosh prokuratura huzuridagi Departamentning Qashqadaryo viloyat boshqarmasi tomonidan bojxona boshqarmasi hamda Farmatsevtika mahsulotlari xavfsizligi markazi Qashqadaryo bo‘linmasi xodimlari bilan hamkorlikda Yakkabog‘ tumani “Janbul” MFY hududida o‘tkazilgan tezkor tadbirda fuqaro S.B. fuqaro A.A.ga “Banksef”, “Zanasef”, “Gansef” va “Gandsef” nomli jami 3 767 dona qalbaki dori vositalarini 54 mln so‘mga sotgan vaqtida ashyoviy dalillar bilan ushlandi.
More
O‘zbekiston Respublikasi Sog‘liqni saqlash vazirligi huzuridagi “Farmatsevtika mahsulotlari xavfsizligi markazi” davlat muassasasi sotuvda "VIPS-MED" MCHJ tomonidan ishlab chiqarilgan, og'iz orqali yuborish uchun suspenziya tayyorlash kukuni shaklidagi "Bar-VIPS" preparatining sertifikatlashtirilmagan 520723 seriyasi aniqlanganligi haqida xabar beradi.
More
Asl Belgisi mobil ilovasidan foydalanib, dori preparatidagi raqamli Data Matrix kodini skanerlash orqali dori vositasi haqida barcha kerakli maʼlumotlarni olishingiz mumkin. Bunday kodda mahsulot tavsifi, qadoq saqlanishi kerak boʼlgan ishlab chiqaruvchi va chakana savdo nuqtasi toʼgʼrisidagi maʼlumotlar va dori vositalari uchun ruxsat beruvchi hujjatlar haqida maʼlumotlar mavjud.
More